What Best Describes the Major Product of an E1 Reaction
In organic chemistry an elimination reaction is a reaction in which two substituents are removed from a molecule to form an alkene. Rate kbase II.
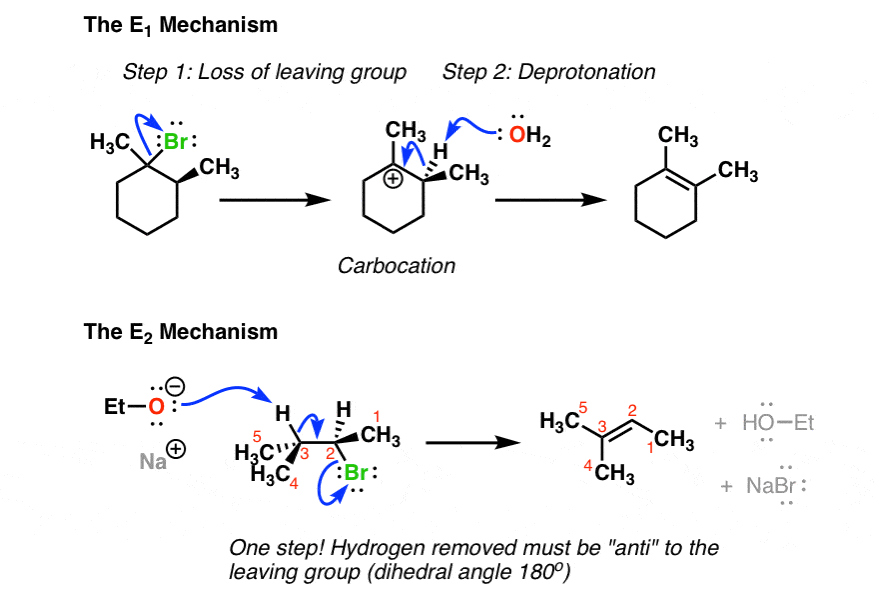
E1 Vs E2 Comparing The E1 And E2 Reactions Master Organic Chemistry
An alkene is a major and only product.

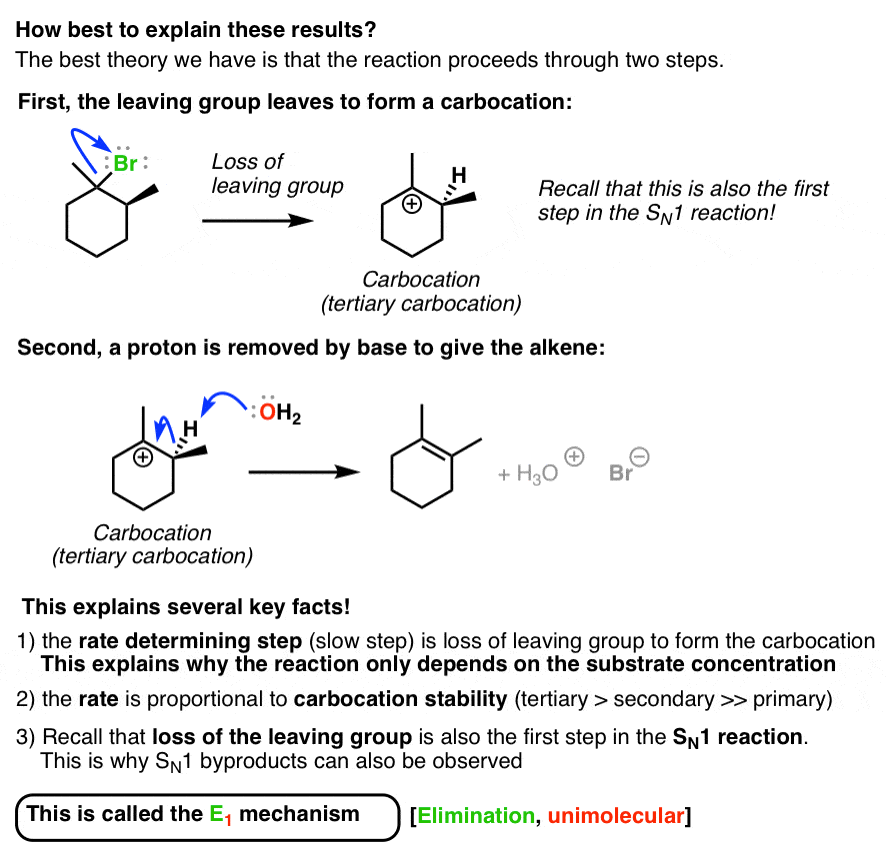
. Rate kRX IV. It is similar to a unimolecular nucleophilic substitution reaction SN1 in particular because the rate determining step involves heterolysis losing the leaving group to form a carbocation intermediate. We look at how regioselectivity and stereoselectivity affect the prediction of the major species in an E1 reaction.
Rearrangements are sometimes seen. More highly alkyl substituted and trans- cis- This E1 mechanistic pathway is. Which of the following statements correctly describes E1 reactions of alkyl halides RX.
A trisubstituted alkene E. Hydroxide removes hydrogen in the β position and these electrons generate a double bond by expelling the leaving group. Stronger bases favor the E1 reaction Which of the following statements about an E1 mechanism is true The identity of the leaving group affects the rate of reaction.
Most often one of them is a hydrogen atom and the other is called the leaving group. Which of the following best describes the stereochemistry of ring closure and the product for the following reaction. E1 reactions usually favour the more stable alkene as the major product.
Predicting the Major Product if an E1 Reaction - Example - YouTube. Rate kbaseRX III. The substituents are usually from adjacent carbons.
Which of the following reactions is predicted to be the fastest in S N 2 mechanism. Elimination reactions of cis- and trans-1-Bromo-2-methylcyclohexanes with N a O E t in E t O H can give the same or different main product 1-methylcyclohexene 1 or 3-methylcyclohexene 2. A monosubstituted alkene D.
No byproducts are obtained here. Rate kRX IV. The major product of the reaction is represented by option B.
-E2 - the major product in the reaction with cyclohexyl bromide with sodium ethoxide in ethanol is formed by this mechanism -E1 - a mechanism that can give a product with a different carbon skeleton from the substrate. Thus the E1 reaction has a relatively early transition state closely. A II and IV B III and V C I.
What would be the major products of the following reaction. Rate kbaseRX III. But many students find one aspect very challenging.
Even though the lower percent yield of products from the E1 path still E1 is a more efficient method for preparing alkenes because E1 is a reversible reaction where it forms alkene via dehydration reaction and forms alcohols via hydration reaction. 84 Which of the following statements correctly describes E1 reactions of alkyl halides RX. What would be the major elimination E1 product of this reaction.
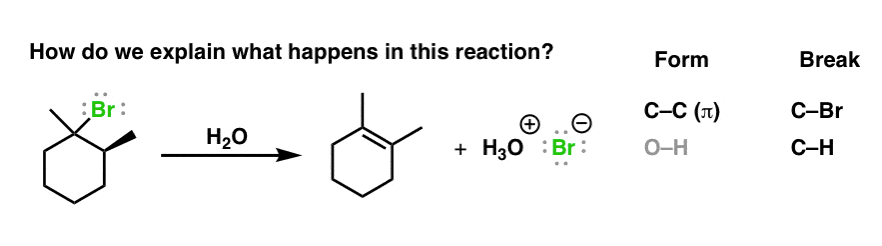
Br is replaced with OH group. The E2 pathway involves a transition state leading from starting material directly to the product. Predicting the Major Product if an E1 Reaction - Example.
Rearrangements are sometimes seen. The product forming step of an E1 reaction is more exothermic than that of an E2 reaction. A II and IV B III and V C I.
A sulfonate ester in the R-configuration B. A sample of 1-chloro-1-phenylethane with an α25D of -948 is reacted with NH3 in methanolwater solvent. E2 and E1 reactions differ significantly in the nature of the transition states that determine the regiochemistry of the product.
Correct option is B SN 2 reaction involves inversion of configuration. Which describes the major product for the reaction shown. Find the value of c for which the following equations have non-trivial solutions.
Sn2 reaction CHCHCONa CHCH3CO q OCCHCH OCCHCH OCCHCH 1 II IV A B II C III D IV E None of these choices. Substitution reaction only but you need to decide whether the mechanism is Sylor Sw2 then choose the. The reactions occur in two or more distinct steps.
E1 and E2 mechanisms give alkenes at the products. As we can see the only. Cx-y-z 0 -cxy-cz 0 and xy-cz 0.
The reactions occur in two or more distinct steps. Because the rate determining. Find The Range Of The Function F 0 1 R F X X 3 X 2 4x 2 Sin 1 X.
The E1 product is favored by an increase in temperature. Unimolecular elimination E1 is a reaction in which the removal of an HX substituent results in the formation of a double bond. Rate kbase II.
If alkene is removed then according to Le Chatelier principle the equilibrium of the reaction will shift to the right resulting. The major substitution product of the reaction is 1-phenyl-1-ethylamine with an α25D of -86. A sulfonate ester in the S-configuration C.
An E2 elimination is a concerted one. Click hereto get an answer to your question What is the major product of the reaction. What would be the major products of the following reaction.
When I teach nucleophilic substitution and elimination reactions I find that students typically have very little trouble drawing each mechanism and predicting the products so long as they are specifically told which reaction. A disrotatory cis-34-diethylcyclobutene B conrotatory cis-34-diethylcyclobutene C disrotatory trans-34-diethylcyclobutene D conrotatory trans-34-diethylcyclobutene 19. Find the value of ab if vectors a and b are given.
What will the major product of an E1 reaction with 2-methyl-3-pentanol be. Find the second excitation energy of Li2. A tetrasubstituted alkene 30.
Click hereto get an answer to your question What is the major product of the reaction. In the E2 reaction we have the same starting compound that is attacked by the base in the first step. Which of a-d indicates the main products.
Predicting the winner of an SN1SN2E1E2 competition.

Electrophilic Aromatic Substitution Mechanism Master Organic Chemistry Organic Chemistry Organic Chemistry Books Organic Chemistry Study
No comments for "What Best Describes the Major Product of an E1 Reaction"
Post a Comment